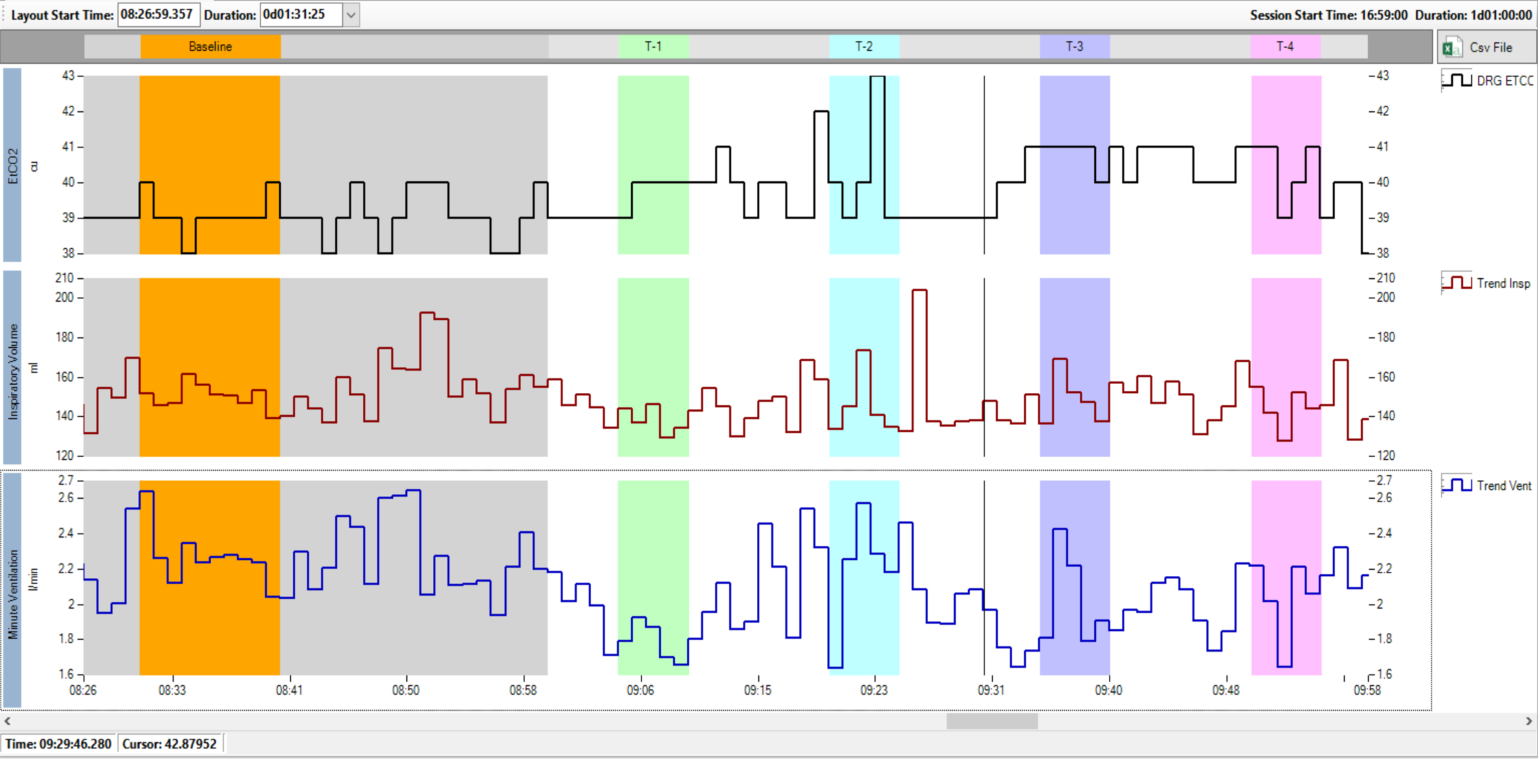
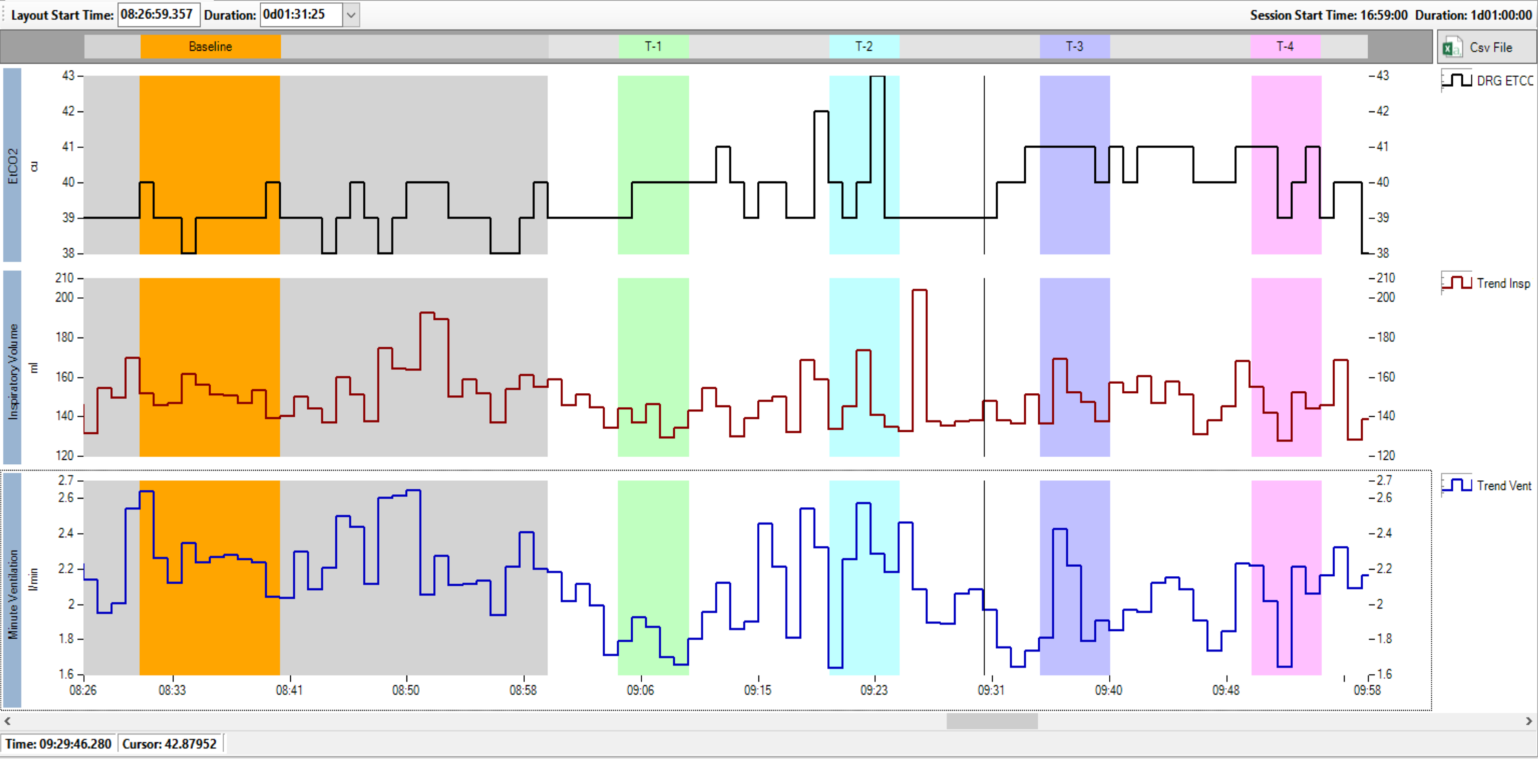
Respiratory insufficiency in post-operative patients following opioid analgesia is a common and life threatening problem. The efficacy of a candidate drug intended to increase minute ventilation by increasing tidal volume and secondarily through increasing respiratory rate (RR) was investigated in a clinical trial. Respiratory ventilation data were collected over several hours using Respiratory Inductance Plethysmography (RIP) using the Nox-T3 device. VivoSense provided custom routines for integration with sedation drug infusion and calibration against pneumotach data. Subjects were awake for several hours of monitoring, including periods of clinical assessments. VivoSense provided objective artifact management and assessments of key respiratory outcome measures.
Dudley Tabakin
Dudley Tabakin, MSc. is Chief Product Officer and co-founder of VivoSense and a fervent believer in “good data” over “big data” in the development of digital endpoints from wearable sensor technology.